Next: The Triplet Relation from Sayre's Equation Up: An Introduction to Direct Methods. The Previous: The -Relation from a Harker-Kasper Inequality
![[IUCr Home Page]](/iucr-top/logos/iucrhome.gif)
![[Commission Home Page]](/iucr-top/logos/cteach.gif)
![]()
![]()
![]()
Next: The Triplet Relation from Sayre's Equation
Up: An Introduction to Direct Methods. The
Previous: The -Relation from a Harker-Kasper Inequality
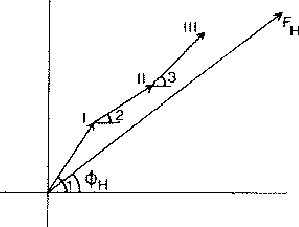
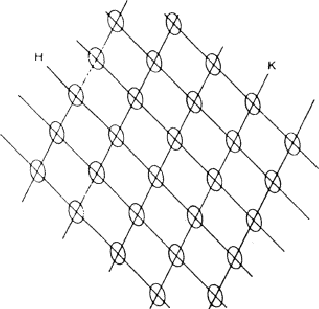
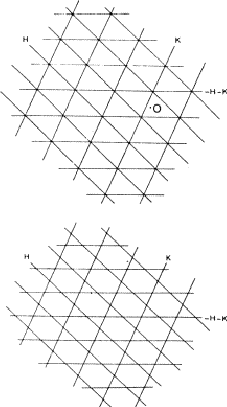
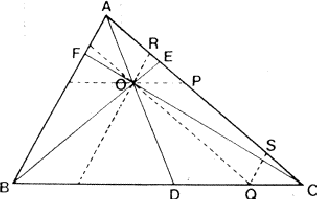
If two reflections H and K are both strong then the electron density is likely to be found in the neighbourhood of the two sets of equidistant planes defined by H and K. That is to say the electron density will be found near the lines of intersection of the planes H and K as indicated in projection in Fig. 9. A large |E| for reflection -H-K as well implies that the electron density will also peak in planes lying d-H-K apart. It is therefore most likely that these planes run through the lines of intersection of the planes H and K, in other words that the three sets of planes have their lines of intersection in common (see Fig. 10a). Then by choosing an origin at an arbitrary point the triplet phase relationship can be found from a planimetric theorem, proved in Fig. 11:
| AO/AD + BO/BE + CO/CF = 2 | (14) |
| (15) |
 |
 |
 |
 |
In Fig. 10a the ideal situation is sketched and of course a small shift of the planes of largest density of -H-K does not affect the reasoning given above. However, the most unlikely position for these planes is the one indicated in Fig. 10b; here the planes -H-K of largest electron density keep clear of the lines of intersection of H and K. The triplet relationship therefore has a probability character and this is emphasised by formulating it as
| (16) |
Copyright © 1984, 1998 International Union of Crystallography
IUCr Webmaster